
Bacterial Communication Crosses into the Anaerobic Frontier
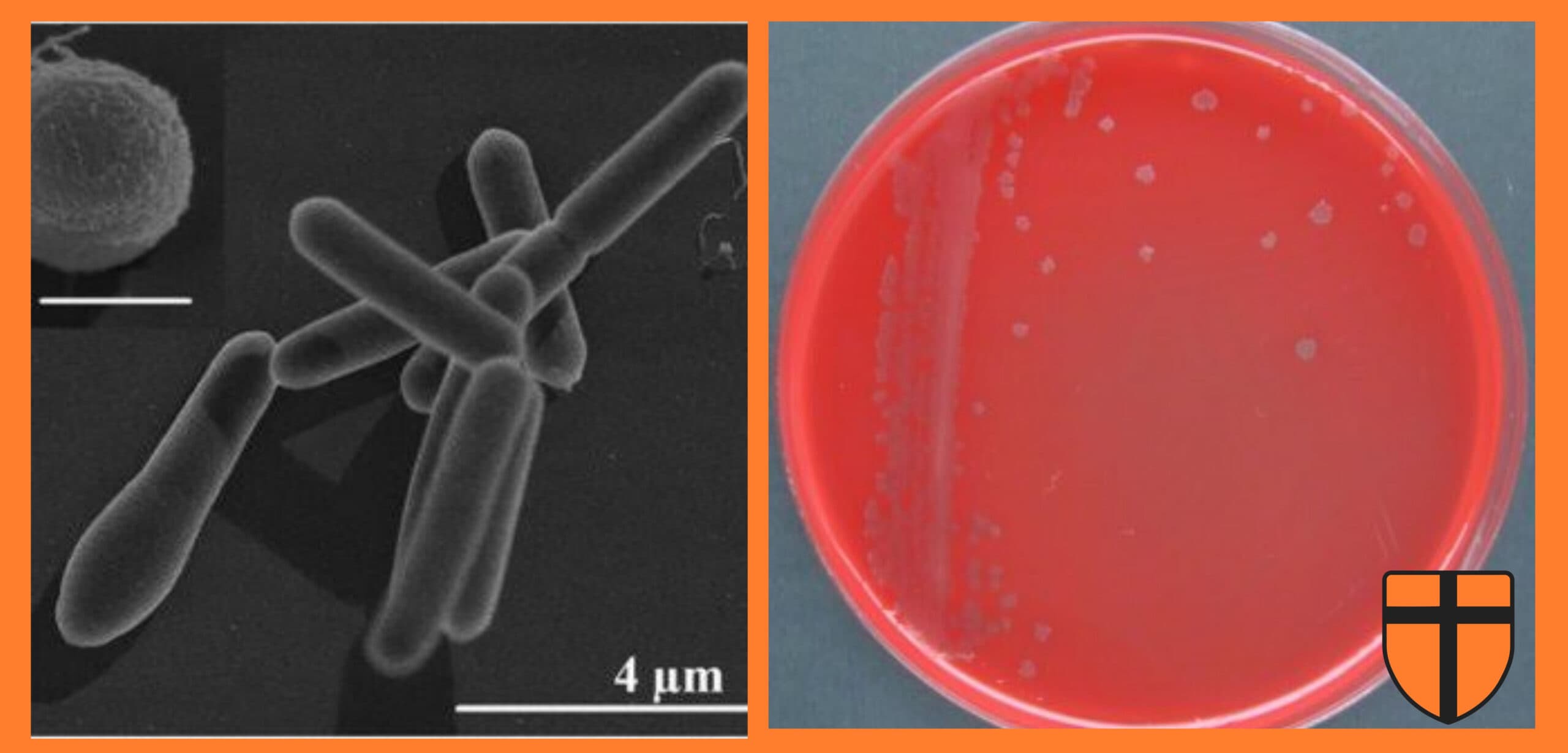
Solid tumors are notoriously difficult to treat. Their cores are oxygen-starved and poorly penetrated by chemotherapy or radiation. But a bacterium called Clostridium sporogenes thrives in exactly these conditions. Its spores remain dormant in healthy tissue and germinate specifically in hypoxic tumor environments. The challenge has been giving it the tools needed to make it a reliable therapeutic agent.
Researchers from the University of Waterloo have taken a significant step toward that goal. For the first time, scientists have installed a functional quorum sensing circuit in an obligate anaerobe. Quorum sensing circuits are genetic switches which activate only when a bacterial population reaches a critical size. Obligate anaerobes are bacteria which live exclusively without oxygen.
Quorum sensing allows bacteria to coordinate behavior based on how many of them are present. Individual cells secrete signaling molecules which accumulate as the population grows. Once that concentration crosses a threshold, it triggers coordinated changes in gene expression across the community. Bacteria, in effect, wait until they have the numbers to act.
This is a valuable property for cancer treatment. Therapeutic genes, those encoding tumor-killing toxins, drug-activating enzymes, or immune-stimulating proteins, could be designed to switch on only after the bacterial population within a tumor reaches a defined size. That would reduce the risk of premature activation in healthy tissue.
Engineering such a system into an obligate anaerobe, however, had never been done. Most genetic tools were developed in oxygen-tolerant organisms, and it was unclear whether a signaling system from one bacterium would function properly in another with very different metabolic constraints.
Writing in ACS Synthetic Biology, Sara Sadr, Bahram Zargar, Marc G. Aucoin, and Brian Ingalls describe how they transplanted a quorum communication system from Staphylococcus aureus into C. sporogenes. The result is a bacterium which can sense its own population density and switch on genes in response, a capability which could make bacterial cancer therapies safer, more precise, and easier to control.
The researchers chose the accessory gene regulator (agr) system from S. aureus, one of the best-studied peptide-based quorum sensing circuits. Rather than small chemical signals, it uses short peptides. Cells secrete a signaling peptide which accumulates outside the cell, and once it reaches sufficient concentration, it triggers a chain reaction which switches on target genes.
To transfer this system into C. sporogenes, the team used a shuttle vector, a genetic vehicle capable of functioning in two different species, to introduce the agr components. They attached a fluorescent protein reporter to the circuit so that activation would produce a visible glow. This allowed them to monitor the system in real time.
The engineered bacteria produced the correct signaling peptide and responded only to that specific signal. The system did not react to unrelated peptides, showing that it retained its natural specificity.
The circuit also behaved exactly as quorum sensing should. It remained inactive when the bacterial population was small, then switched on sharply once the culture reached a critical density. At that point, gene activity increased by about 2.6-fold per cell.
Further tests showed that activation depended on the buildup of the signaling peptide in the surrounding environment. When that accumulated signal was removed, activation was delayed and reduced. Together, these results demonstrate that the transplanted circuit functioned as a true, density-dependent communication system inside the anaerobic bacterium.
The discovery shows great medical potential. C. sporogenes already colonizes tumor cores selectively. A programmable quorum circuit means therapeutic genes can now be set to activate at a defined population threshold, adding a layer of precision which was previously unavailable in anaerobic bacterial therapeutics.
The implications extend to industry as well. Clostridial species are widely used in fermentation for solvent production and specialty chemicals, including butanol and 3-indolepropionic acid, a compound with antioxidant and neuroprotective properties. In fermentation, activating production pathways too early can slow growth and reduce yield. A density-dependent switch allows cells to build biomass first and begin producing compounds only once they reach sufficient numbers.
This study is the first successful reconstruction of a peptide-based quorum sensing system in an obligate anaerobe. It shows that these communication circuits can function outside their original host, even in organisms with very different metabolic environments.
In synthetic biology, expanding the range of programmable organisms is as important as refining the circuits themselves. Bacterial communication has now crossed into the anaerobic frontier.





