
Tiny Lab-Grown Spinal Cords Could Transform How We Treat Paralysis
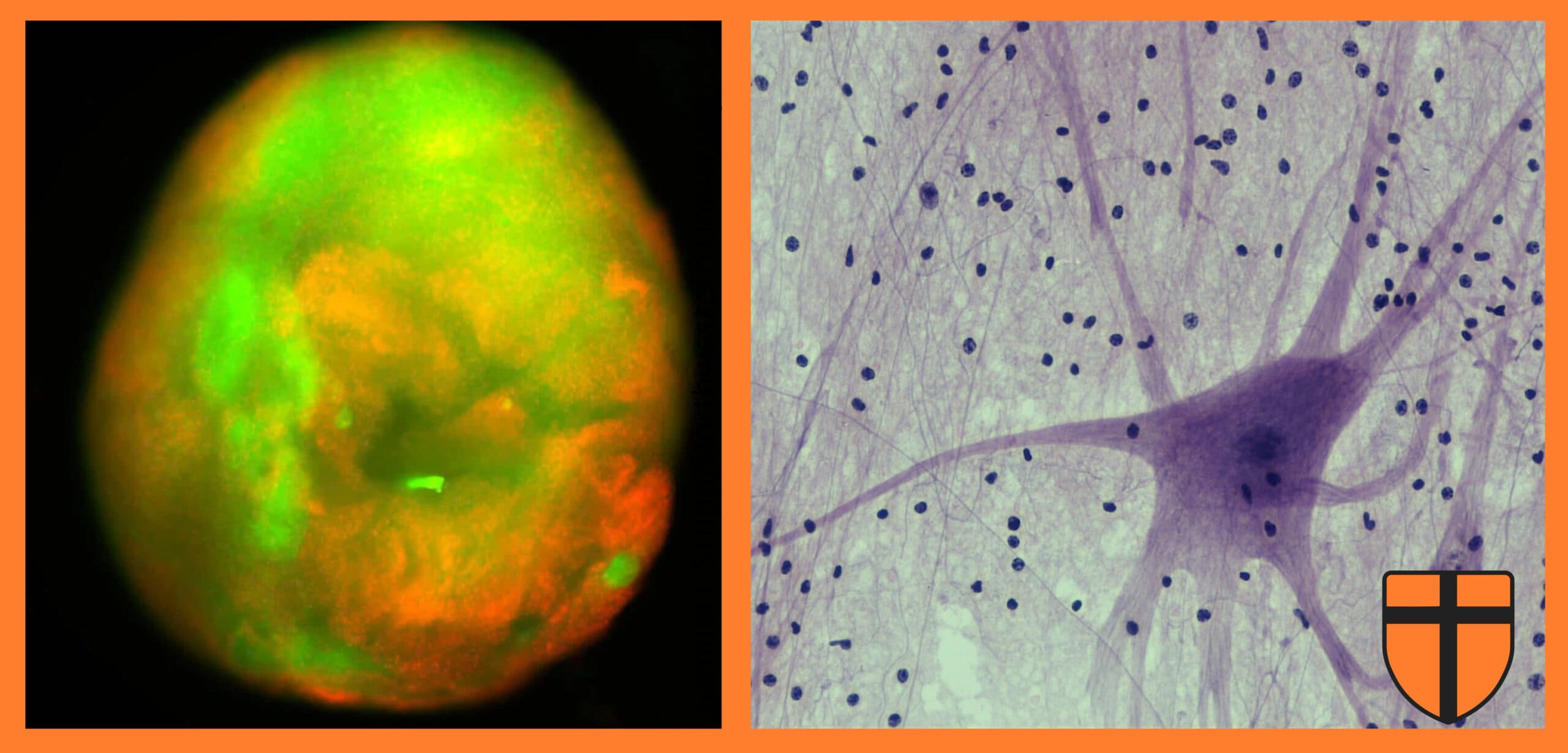
In a recent study published in Nature Biomedical Engineering, researchers report a sophisticated new platform for studying spinal cord injury (SCI): three-dimensional human spinal cord organoids grown from stem cells. These miniature, lab-grown tissues replicate key structural and cellular features of the human spinal cord and can be experimentally injured and treated under controlled conditions. The approach offers a promising alternative to traditional animal models and may accelerate the development of therapies for paralysis.
Spinal cord injury remains one of the most challenging conditions in regenerative medicine. An estimated 250,000 to 500,000 people worldwide sustain an SCI each year. Beyond the initial mechanical trauma, injuries trigger a complex cascade of secondary damage, including inflammation, widespread cell death, and the formation of a glial scar. This scar arises when astrocytes, normally supportive cells, densely accumulate around the injury site. While protective in the short term, the scar creates both physical and chemical barriers which inhibit axonal regrowth, severely limiting functional recovery.
To model these processes, the researchers generated spinal cord organoids which replicate the cellular diversity of human tissue. They subjected the organoids to two clinically relevant forms of injury. A scalpel-induced laceration simulated penetrating trauma, while a compressive contusion reproduced the blunt-force damage most commonly observed in patients. Cellular injury was quantified using fluorescent viability dyes which distinguish living from dead cells, along with assays measuring lactate dehydrogenase, an enzyme released when cells rupture. Together, these methods allowed precise assessment of tissue damage and repair.
A central innovation of the study was the application of bioactive peptide amphiphile nanofibres. These molecules spontaneously self-assemble into filaments thousands of times thinner than a human hair, forming a hydrogel when introduced into tissue. Hydrogels are water-rich polymer networks which resemble the extracellular matrix, the structural scaffold which surrounds and supports cells in living organisms. By mimicking this natural environment, hydrogels can provide both mechanical support and biochemical cues which influence cellular behavior.
The researchers focused on nanofibres displaying the IKVAV peptide sequence, derived from laminin-1, a protein abundant in neural tissue. Previous work in mouse models of SCI has shown that IKVAV-containing materials promote neuronal growth. In the organoid system, the bioactive hydrogel significantly reduced glial scar-like tissue and enhanced axonal regeneration, the extension of neurons’ long projections responsible for transmitting electrical signals. These findings align with a broader body of biomaterials research demonstrating that engineered scaffolds can modulate inflammation, reduce fibrotic tissue formation, and encourage neural repair.
Recognizing that inflammation plays a central role in SCI pathology, the team further refined their model by introducing microglia, the resident immune cells of the central nervous system. These immune-competent organoids more closely mirrored the post-injury environment of human tissue. Following injury, the organoids exhibited increased expression of complement component C1QA, a marker of reactive microglia, and complement component C3, associated with a neurotoxic subtype of reactive astrocytes. Both markers are characteristic of inflammatory responses observed in vivo.
Transcriptomic analysis revealed that treatment with the bioactive peptide amphiphile hydrogel reduced expression of key pro-inflammatory genes, including TNF-α and IL-1β. These cytokines, when overproduced after injury, amplify tissue destruction and inhibit regeneration. By dampening this inflammatory signaling, the hydrogel appears not only to support structural repair but also to shift immune activity toward a more regenerative state. Such immunomodulation has emerged as a critical objective in contemporary SCI therapy development.
The integration of human organoids with programmable biomaterial scaffolds represents a significant advance in translational neuroscience. Organoid models enable mechanistic studies in human-derived tissue while reducing dependence on animal experimentation. At the same time, biomaterials such as peptide amphiphile hydrogels can be systematically tuned to optimize mechanical properties, molecular signaling, and immune responses.
Important challenges remain before such strategies can be translated to clinical practice. Organoids lack vascular networks and full systemic immune interactions, and scaling biomaterial therapies for safe use in patients will require rigorous testing. Nonetheless, the study demonstrates a powerful experimental framework for dissecting the interplay among inflammation, scarring, and axonal disruption in spinal cord injury.
As regenerative medicine moves toward increasingly human-specific models, the convergence of stem cell biology and bioengineered scaffolds may help overcome longstanding barriers in neural repair. With continued refinement, these approaches could bring the field closer to effective therapies which restore mobility and improve quality of life for individuals living with spinal cord injury.





